CONTACT & STAFF
Facility Tel.: +55 19 3518 2349
Facility E-mail: caterete@lnls.br
Coordination: Florian E. P. Meneau
Tel.: +55 19 3512 1132
E-mail: florian.meneau@lnls.br
Click here for more information on this Facility team.
The CATERETÊ beamline provides unique capabilities in biological and condensed matter imaging and dynamics experiments with particular focus on the application of coherent X-ray scattering and diffraction techniques. Coherent X-ray diffractive imaging (CXDI) and X-ray photon correlation spectroscopy (XPCS) experiments are the heart of the activities at the CATERETÊ beamline, but also time-resolved small angle X-ray scattering (SAXS) and Ultra-Small Angle (USAXS), which will benefit of the high flux of the source.
The beamline is equipped with an undulator source, in a low-beta straight section, and two cryo-cooled focussing mirrors creating a 30 x 30 µm2 (FWHM) fully coherent beam at the sample position. The beamline operates in the 3 to 24 keV using a horizontally deflecting 4-bounce crystal monochromator (4CM). Moving the 4CM laterally by a few mm, enables to operate the beamline in pink beam mode (ΔE/E = 1-2%), maintaining the beam position unchanged. The experimental station is located 88 m from the source, followed by a 28 meters vacuum chamber hosting the Medipix (3k x 3k pixels2) in-vacuum detector. The sample-to-detector distance can be adjusted between 1 and 28m.
The CATERETÊ beamline will make possible to answer diverse and multiple questions related to life sciences (biological and medical applications), structural biology (proteins, lipids, macromolecules) and the vast field of material sciences including nanotechnology (energy, food and health, photonics), polymer sciences, catalysis, rheology and environmental sciences (geosciences, oil prospection, catalysis). Therefore, all major research fields, from physics, chemistry and biology, but as well industrial aspects can be explored on CATERETÊ.
Facility Tel.: +55 19 3518 2349
Facility E-mail: caterete@lnls.br
Coordination: Florian E. P. Meneau
Tel.: +55 19 3512 1132
E-mail: florian.meneau@lnls.br
Click here for more information on this Facility team.
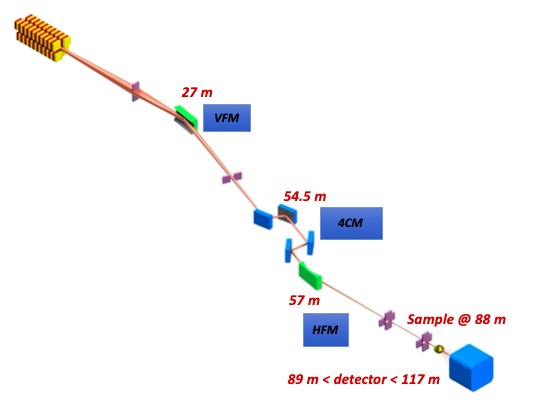
| Element | Type | Position [m] | Description |
|---|---|---|---|
| Source | Kyma APU | 0 | 22mm period (51) |
| VFM | Vertical focusing mirror | 27 | |
| Mono | 4 crystal monochormator | 56 | Fixed exit, double channel-cut |
| HFM | Horizontal focusing mirror | 57 | |
| Sample | 88 | ||
| Detector (SAXS) | Medipix | 90-118 | 3000 x 3000 pixels² Medipix3 PITEC |
| Detector (WAXS) | Pilatus 300K | 0.3-1.5 |
| Parameter | Value | Condition |
|---|---|---|
| Energy Range | 3 – 24 keV | |
| Coherence flux | > 10 11 ph/s @ 6 keV
> 10 10 ph/s @ 9 keV
|
|
| Beam size at sample | 1 – 30 µm | |
| Sample to detector distance | up to 30 m |
CXDI is a high resolution, lensless-imaging technique in which an image is obtained from the X-ray diffraction pattern. An isolated object is illuminated with coherent X-rays and the resulting diffraction pattern is recorded in the far field. Then, a phase retrieval algorithm is applied to recover the phase and obtain the image in real space. CXDI can reveal the complex 3-dimensional morphology of a wide variety of crystalline and non-crystalline materials with nanometric resolution.
This technique allows the investigation of dynamics on nanometric scale based on the observation of fluctuations in the intensity of the coherent X-ray speckle pattern over time. XPCS is ideally suited for probing structural dynamics on a variety of nanometer length scales and over a time span ranging from milliseconds to hundreds of seconds and is suitable for the study of soft and hard condensed matter.
These techniques allow the determination of structural parameters such as molecular distances, radius of gyration, pore sizes, morphology, state of aggregation etc., with dimensions ranging from ångström to several micrometers. In addition, it is possible to perform time resolved SAXS and USAXS to follow changes in size and property in soft matter and biological systems with time-resolution from sub-milliseconds up to minutes. The combination of SAXS/USAXS allows the elucidation of static and transitory hierarchical structures in soft condensed matter, biophysical systems and non-crystalline biological structures.

Cryogenic sample environment for CXDI at Cateretê beamline
Ptychography is available for 2D and 3D imaging at 3.8 and 10 keV (pink beam (ΔE/E = 1-2%)). Samples are on silicon nitride membranes and aligned with an on-axis microscope. The setup is compatible with a cryo-system which is still under commissioning and will be available soon. 2D and 3D reconstructions can be retrieved by an in-house software.
XPCS can be performed in SAXS geometry at 10 keV (pink beam (ΔE/E = 1%)) with up to 28 m sample to detector distance. In-air sample stage for capillaries and wafers are available, a heating stage is under commissioning and will be available soon. XPCS data analysis can be performed by an in-house python code.
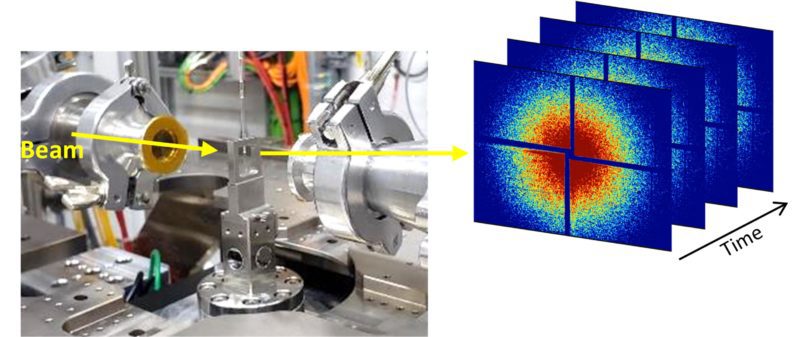
XPCS measurements of colloidal gold particles at Cateretê beamline
We encourage users to contact the beamline staff for more information on sample preparation and environment.
For CXDI measurements, the samples are supported on 100 nm-thick silicon nitride membranes. We recommend the following:
Radiation sensitive samples that require cryogenic protection can be prepared onsite. We are equipped with a cryostat (Leica CM1860UV5337) for sample micro-sectioning at low temperatures ( -30°C) and a fast-freezing (EMS Plunge-freezer) device to cool the samples down to cryogenic temperature. A laser microdissection microscope (Leica LMD7), with micrometric precision and a microtome (Leica HistoCore AUTOCUT) for paraffin and resin embedded samples micro-sectioning are available. The users interested to use this equipment should contact the beamline staff and include this information in the proposal.
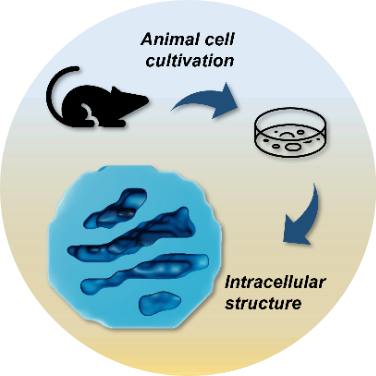
The comprehension of the tridimensional intracellular structures and cellular morphology are key elements to understand physiological states and those related to world affecting diseases. Coherent X-ray imaging can provide quantitative information regarding large volume specimens such as tissue fragments (tens of micrometers) and eukaryotic cells down to tens of nanometers resolution. Similar experiments performed by our group:
Polo, C.C., Fonseca-Alaniz, M.H., Chen, JH. et al. Three-dimensional imaging of mitochondrial cristae complexity using cryo-soft X-ray tomography. Sci Rep 10, 21045 (2020). DOI: 10.1038/s41598-020-78150-3
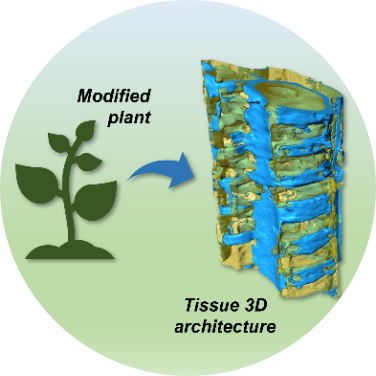
Plant genetic manipulation technology can improve biomass digestibility for biofuel and biotechnological sectors. High resolution 3D imaging can provide quantitative morphological information about the cellular wall anatomical architecture. This can help to understand the generated phenotypes from mutant plants and evaluate their applicability to large scale cultivation. Related experiments performed by our group:
Polo, C.C., Pereira, L., Mazzafera, P. et al. Correlations between lignin content and structural robustness in plants revealed by X-ray ptychography. Sci Rep 10, 6023 (2020). DOI: 10.1038/s41598-020-63093-6
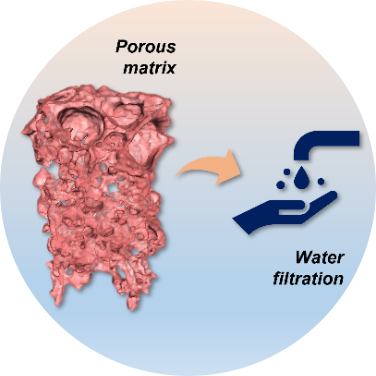
The porosity and complex morphology of porous materials and membranes drive their applications in challenging separations varying from biomolecules purification to catalysts recovery in the chemical industry, gas and vapor separation in oil refineries, pollutant particle filtration for air purification. A 3D high-resolution imaging of a representative membrane volume would enable a much more comprehensive representation of the porous systems, help to better understand transport phenomena, the structure functioning and its formation mechanism, and guide future developments.
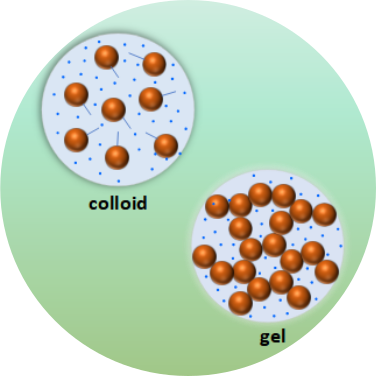
Soft matter displays complex dynamics ranging from nanometers to micrometers and timescales from miliseconds to minutes. The understanding of the dynamical properties in soft matter as colloids, gel, proteins, polymers, and complex liquids can help address challenging scientific problems such as aggregation, stability, phase separation and unravel hidden mechanisms in these complex systems. XPCS is ideally suited for probing dynamics in non-equilibrium systems not accessible with most scanning probes, even for colored and opaque systems which is particularly well matched for the study of disordered soft materials.